Originally posted 11 June 2015
Updated 16 September 2023
My dad
That’s my dad. No, not the dapper gentleman standing in the back. He is the diapered baby sitting in front. One interesting thing about this circa 1924 photograph, I can tell you unequivocally, is that both males in the photograph are carriers of 22q13 deletion syndrome. I can say this even though neither man was ever genetically tested and neither ever had children with a known diagnosis of 22q13 deletion syndrome. How can I be so sure? I spent several years contacting relative and having them tested. It was an interesting and often challenging undertaking. My motivation was to warn family members and at least prepare them for the possibility of having to raise a son or daughter with 22q13 deletion syndrome. Along the way I received “thank you” from some family members and angry words from others. Some family members simply did not want to know. It is a story for some future blog, I suppose.
From this family research I was able to deduce that Joe, my dapper grandfather, was a carrier of 22q13 deletion syndrome with a “balanced translocation” of genetic material between chromosome 22 and chromosome 19. Some of my grandfather’s siblings were carriers and at least two of Joe’s sons were carriers. My dad was one of the carriers. He had four sons, including me, and I am a carrier. My dad did not have children with 22q13 deletion syndrome, but I had two.
The power of genetic principles
I know my dad was the carrier (not my mom) because a few of dad’s relatives are carriers. (See the line chart on my page “Who is arm 22q13?“.) I was able show that my grandfather was a carrier using similar family evidence. Genetics and inheritance follow certain rules and those rules can be used to peer into the past. Genetics and evolution are two different aspects of the same rules, and understanding them can be very powerful tools for understanding where we come from and where we might be going.
Somewhere between 15% and 24% of all children with terminal deletions inherit that deletion from a carrier parent. If your family has carriers, nature has provided a curious way to remove carriers from future generations: have small families. This graph shows why.
(right click on graph to enlarge in a new window)
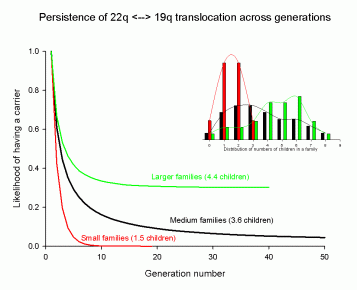 The main graph has three colored lines. (Ignore the small “inset” graph with bars; it provides details some researchers might want to see.) The green line on the main graph represents what happens when people in the extended family have relatively large families (4.4 children, on average). The black line shows the same process when the average family size is less (3.6 children per family). The red line shows the impact of small families (1.5 children per family, on average). What impact are we talking about? The beginning of the graph starts today. The end of the graph shows what happens after 10 to 50 generations from today. Since most people assume 25 years for each generation to pass, the first 10 generations will take 250 years. Here is the point. If people have only small families, we can expect carriers to disappear (reach 0.0 on the scale) from the population in fewer than 10 generations. However, if people choose to have large families (green line), carriers are unlikely to ever disappear (green line never reaches zero).
The main graph has three colored lines. (Ignore the small “inset” graph with bars; it provides details some researchers might want to see.) The green line on the main graph represents what happens when people in the extended family have relatively large families (4.4 children, on average). The black line shows the same process when the average family size is less (3.6 children per family). The red line shows the impact of small families (1.5 children per family, on average). What impact are we talking about? The beginning of the graph starts today. The end of the graph shows what happens after 10 to 50 generations from today. Since most people assume 25 years for each generation to pass, the first 10 generations will take 250 years. Here is the point. If people have only small families, we can expect carriers to disappear (reach 0.0 on the scale) from the population in fewer than 10 generations. However, if people choose to have large families (green line), carriers are unlikely to ever disappear (green line never reaches zero).
Let me be clear. I am not advocating for any specific choice. This is not about ethics. In a sense, these are God’s rules. They are inferred from the statistics of inheritance in the same way quantum tunneling is inferred from the statistics of nuclear emission. I worked with a member of my family to generate this graph using a mathematical simulation. I wanted to know how long 22q13 deletion syndrome has been in our family. The answer comes from the green line. Historically, my European ancestors had large families. My great-grandfather had six children. His children had an average of 4.8 children each. These numbers suggest that the translocation could have existed in our family for tens of generations.
arm
